RESEARCH
RESEARCH
Materials Electrochemistry for Energy and Sustainability
OVERVIEW
Renewable energy sources, such as solar and wind, have huge potential to minimize our dependence on fossil fuels and to reduce greenhouse gas emission. However, photovoltaics and wind are uncertain and intermittent, which create grand challenges to the traditional electricity grid at all levels. In order to make renewable energy a major contribution in the future energy portfolio (available whenever and wherever it is needed), it is important to develop efficient and cost effective solutions to energy storage and conversion. Storing grid electricity in batteries or catalytically converting electrical energy to renewable fuels and chemicals, such as hydrocarbons, alcohols, and ammonium, can overcome the mismatch between renewable energy production and demand. The development of these technologies requires efficient electrochemical systems that can operate at appropriate temperatures with minimal energy losses. We need to make revolutionary advances in materials chemistry, discovering new materials, syntheses, and functionalities. Therefore, our research is positioned at the crossroads of interdisciplinary energy research, with the ultimate goal of achieving unprecedented control of materials properties for next-generation energy applications as well as contributing to the fundamental progress of materials electrochemistry and solid state chemistry.
Our research covers the design and synthesis, processing, characterization, and applications of energy materials in electrochemical systems (batteries, catalysis, and smart windows). To accelerate the materials discovery, we develop an integral analytical program to study materials dynamics under operating conditions using operando synchrotron X-ray and electron analytical techniques. Our team collaborates with a number of Department of Energy National Laboratories, domestic and international institutions, and companies.
OVERVIEW
Renewable energy sources, such as solar and wind, have huge potential to minimize our dependence on fossil fuels and to reduce greenhouse gas emission. However, photovoltaics and wind are uncertain and intermittent, which create grand challenges to the traditional electricity grid at all levels. In order to make renewable energy a major contribution in the future energy portfolio (available whenever and wherever it is needed), it is important to develop efficient and cost effective solutions to energy storage and conversion. Storing grid electricity in batteries or catalytically converting electrical energy to renewable fuels and chemicals, such as hydrocarbons, alcohols, and ammonium, can overcome the mismatch between renewable energy production and demand. The development of these technologies requires efficient electrochemical systems that can operate at appropriate temperatures with minimal energy losses. We need to make revolutionary advances in materials chemistry, discovering new materials, syntheses, and functionalities. Therefore, our research is positioned at the crossroads of interdisciplinary energy research, with the ultimate goal of achieving unprecedented control of materials properties for next-generation energy applications as well as contributing to the fundamental progress of materials electrochemistry and solid state chemistry.
Our research covers the design and synthesis, processing, characterization, and applications of energy materials in electrochemical systems (batteries, catalysis, and smart windows). To accelerate the materials discovery, we develop an integral analytical program to study materials dynamics under operating conditions using operando synchrotron X-ray and electron analytical techniques. Our team collaborates with a number of Department of Energy National Laboratories, domestic and international institutions, and companies.
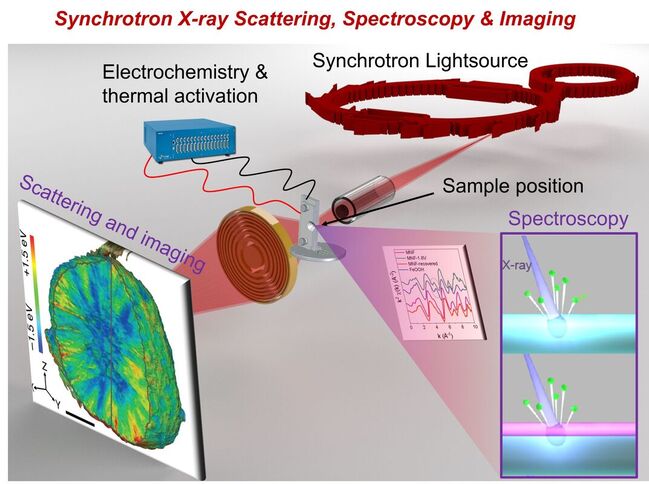
Synchrotron X-ray techniques stand out as one of the most effective methods for the multiscale probing of electronic and geometric structures through spectroscopy, scattering, and imaging capabilities Compared with the laboratory X-ray sources, the advantage of synchrotron radiation includes the continuously tunable photon energy across a wide range with pertained high brightness and flux. Beyond designing and synthesizing advanced energy materials in the Lin Lab, we also make use of synchrotron facilities at different DOE national labs, including Stanford Synchrotron Radiation Lightsource at SLAC, Advanced Light Source at LBNL, National Synchrotron Light Source II at BNL, and Advanced Photon Source at Argonne. We also collaborate with synchrotron facilities in other countries, e.g., European Synchrotron Radiation Facility.
RESEARCH THRUSTS
Thrust 1. Controlling Phase Transformations, Crystallographic Defects, Charge Distribution, and Chemomechanical Properties to Design Better Batteries
Representative publications
L. Mu, F. Lin* et al. Nat. Commun. 9, 2810 (2018); L. Mu, F. Lin* et al. Nano Lett. 18, 5, 3241 (2018); Z. Jiang, F. Lin* et al. Nat. Commun. 11, 2310 (2020); Y. Yang, F. Lin* et al. Adv. Energy Mater. 9, 1900674 (2019); Z. Xu, F. Lin* et al. Nat. Commun. 11, 83 (2020); Z. Xu, F. Lin* et al. Adv. Mater. 2003417 (2020); S. Spence, F. Lin* et al. Inorg. Chem. 59, 10591 (2020); M. M. Rahman, F. Lin* et al. Nat. Commun. 11, 4548 (2020); F. Lin* et al. Chem. Rev. 117, 13123 (2017); M. M. Rahman, F. Lin* et al. Energy Environ Sci. 11, 2496 (2018); Y. Sun, F. Lin* et al. Nano Lett. 21, 4570 (2021); J. Li, F Lin* et al. Science 376, 517-521 (2022).
Representative publications
L. Mu, F. Lin* et al. Nat. Commun. 9, 2810 (2018); L. Mu, F. Lin* et al. Nano Lett. 18, 5, 3241 (2018); Z. Jiang, F. Lin* et al. Nat. Commun. 11, 2310 (2020); Y. Yang, F. Lin* et al. Adv. Energy Mater. 9, 1900674 (2019); Z. Xu, F. Lin* et al. Nat. Commun. 11, 83 (2020); Z. Xu, F. Lin* et al. Adv. Mater. 2003417 (2020); S. Spence, F. Lin* et al. Inorg. Chem. 59, 10591 (2020); M. M. Rahman, F. Lin* et al. Nat. Commun. 11, 4548 (2020); F. Lin* et al. Chem. Rev. 117, 13123 (2017); M. M. Rahman, F. Lin* et al. Energy Environ Sci. 11, 2496 (2018); Y. Sun, F. Lin* et al. Nano Lett. 21, 4570 (2021); J. Li, F Lin* et al. Science 376, 517-521 (2022).
Background and Scientific Impacts
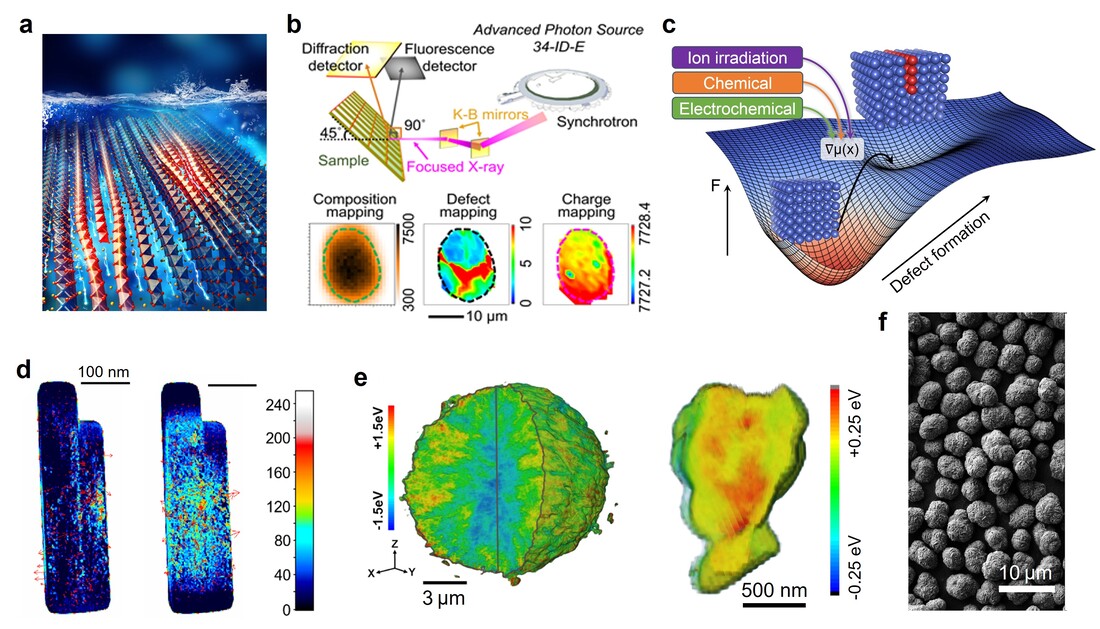
Chemical and mechanical properties interact on the nanometric scale and collectively govern the functionalities of ion-intercalating solids. Understanding the relationship between the two can inform the synthesis of electrode materials with optimal chemomechanical properties for long-life, high-energy batteries. Our goal is to understand the defect inception, accumulation, and growth at the interface or within the grains, as well as to establish the defect-microstructure-performance relationship for oxide cathodes using synchrotron X-ray analyses, electrochemical methods, and data mining. Thus far, our team has made seminal contributions to developing methods for multiscale mapping of phase transformations, defects, charge distribution, and chemomechanical properties as well as elucidated the design rules for synthesizing layered oxides that are resilient against chemomechanical breakdown. We have developed an integrative approach of mapping valence states and constructing chemical topographies to investigate the redox phase transformation in polycrystalline layered oxide cathode materials under thermal abuse conditions. Using in situ transmission electron microscopy, we have reported a mechanism of nanoscale chemomechanical breakdown in layered oxide cathode materials, originating from oxygen release at high states of charge under thermal abuse conditions. Through Laue X-ray microdiffraction, many crystallographic defects are spatially identified and statistically quantified from a large quantity of diffraction patterns in many layered oxide particles, including geometrically necessary dislocations, tilt boundaries, and mixed defects. Our in situ and ex situ measurements, combining microdiffraction and X-ray spectroscopic imaging, reveal that layered cathode particles with a higher concentration of geometrically necessary dislocations provide deeper charging reactions, indicating that dislocations may facilitate redox reactions in layered oxides at low states of charge. We have also elucidated the spatially resolved charge distribution in lithium layered oxides with different grain crystallographic arrangements and established a mathematical model to quantify their charge distributions. While the holistic “surface-to-bulk” charge distribution prevails in polycrystalline particles, the crystallographic orientation-guided redox reaction governs the charge distribution in the local charged nanodomains. Compared to the randomly oriented grains, the radially aligned grains exhibit a lower cell polarization and higher capacity retention upon battery cycling. The radially aligned grains create less tortuous lithium ion pathways, thus improving the charge homogeneity as statistically quantified from over 20 million nanodomains in polycrystalline particles.
Chemical and mechanical properties interact on the nanometric scale and collectively govern the functionalities of ion-intercalating solids. Understanding the relationship between the two can inform the synthesis of electrode materials with optimal chemomechanical properties for long-life, high-energy batteries. Our goal is to understand the defect inception, accumulation, and growth at the interface or within the grains, as well as to establish the defect-microstructure-performance relationship for oxide cathodes using synchrotron X-ray analyses, electrochemical methods, and data mining. Thus far, our team has made seminal contributions to developing methods for multiscale mapping of phase transformations, defects, charge distribution, and chemomechanical properties as well as elucidated the design rules for synthesizing layered oxides that are resilient against chemomechanical breakdown. We have developed an integrative approach of mapping valence states and constructing chemical topographies to investigate the redox phase transformation in polycrystalline layered oxide cathode materials under thermal abuse conditions. Using in situ transmission electron microscopy, we have reported a mechanism of nanoscale chemomechanical breakdown in layered oxide cathode materials, originating from oxygen release at high states of charge under thermal abuse conditions. Through Laue X-ray microdiffraction, many crystallographic defects are spatially identified and statistically quantified from a large quantity of diffraction patterns in many layered oxide particles, including geometrically necessary dislocations, tilt boundaries, and mixed defects. Our in situ and ex situ measurements, combining microdiffraction and X-ray spectroscopic imaging, reveal that layered cathode particles with a higher concentration of geometrically necessary dislocations provide deeper charging reactions, indicating that dislocations may facilitate redox reactions in layered oxides at low states of charge. We have also elucidated the spatially resolved charge distribution in lithium layered oxides with different grain crystallographic arrangements and established a mathematical model to quantify their charge distributions. While the holistic “surface-to-bulk” charge distribution prevails in polycrystalline particles, the crystallographic orientation-guided redox reaction governs the charge distribution in the local charged nanodomains. Compared to the randomly oriented grains, the radially aligned grains exhibit a lower cell polarization and higher capacity retention upon battery cycling. The radially aligned grains create less tortuous lithium ion pathways, thus improving the charge homogeneity as statistically quantified from over 20 million nanodomains in polycrystalline particles.
Thrust 2. Molecular Level Investigation of Solid–liquid Interfaces in Electrochemical Energy Systems
Representative publications: C. Kuai, F. Lin* et al. Nature Catalysis 3, 743 (2020); J. Steiner, F. Lin* et al ACS Appl. Mater. Interface 11, 37885 (2019); C. Kuai, F. Lin* et al. ACS Catal. 9, 6027 (2019); L. Mu, F. Lin* et al. Chem. Mater. 31, 9769 (2019); L. Mu, F. Lin* et al. Adv. Energy Mater. 8, 1801975 (2018); C. Kuai, F. Lin* et al. JACS (2021); Z. Yang, F. Lin* et al. Adv. Energy Mater. 11, 2002719 (2021); F. Lin* et al. Chem. Rev. 117, 13123 (2017); A. Hu, F. Lin* et al. J. Mater. Chem. A 8, 20000 (2020).
Representative publications: C. Kuai, F. Lin* et al. Nature Catalysis 3, 743 (2020); J. Steiner, F. Lin* et al ACS Appl. Mater. Interface 11, 37885 (2019); C. Kuai, F. Lin* et al. ACS Catal. 9, 6027 (2019); L. Mu, F. Lin* et al. Chem. Mater. 31, 9769 (2019); L. Mu, F. Lin* et al. Adv. Energy Mater. 8, 1801975 (2018); C. Kuai, F. Lin* et al. JACS (2021); Z. Yang, F. Lin* et al. Adv. Energy Mater. 11, 2002719 (2021); F. Lin* et al. Chem. Rev. 117, 13123 (2017); A. Hu, F. Lin* et al. J. Mater. Chem. A 8, 20000 (2020).
Background and Scientific Impacts
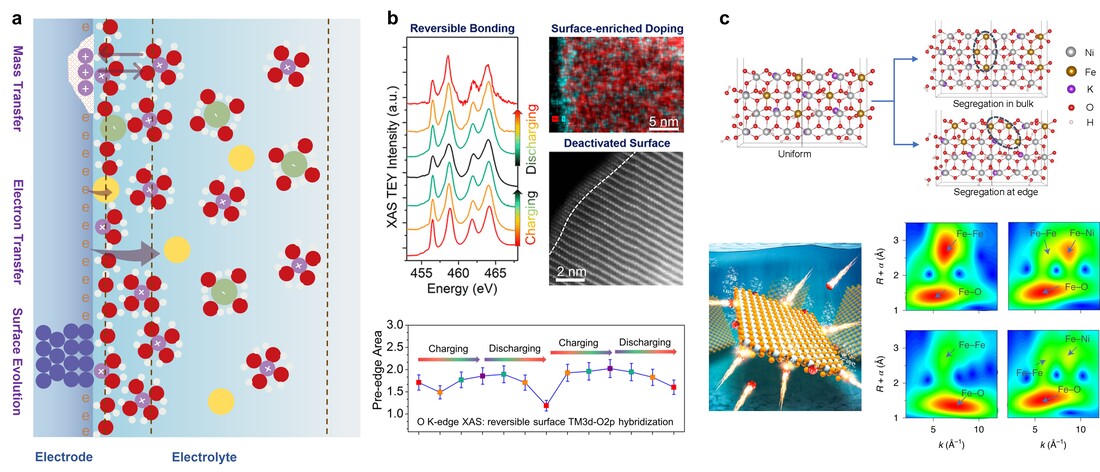
The solid–liquid interfacial reactivity and stability govern the efficiency and lifetime of electrochemical energy devices. Probing and improving the electrochemical solid–liquid interface under operating conditions represents frontier challenges in many aspects of energy sciences, including but not limited to electrocatalytic processes, energy storage and conversion, and chemical synthesis. Our research team has undertaken significant research activities to modulate the electrode surface chemistry for enhancing the interfacial reactivity in batteries and electrocatalysis. Our studies have also found that the high interfacial reactivity triggers undesired reactions with the electrolyte leading to structural changes at the electrode surface and ultimately performance degradation. The mass and charge transfer across the solid–liquid electrochemical interface creates a dynamic environment (e.g., ion concentration) that is sensitive to the electrochemical protocol, electrode surface chemistry, and electrolyte composition. Understanding the interplay between electrode surface chemistry and electrolyte composition will inform the design of better electrochemical interfaces at the atomic and molecular scales. However, probing and tailoring the solid–liquid interfacial processes has ubiquitously remained a key challenge in electrochemical systems. Our long-term goal is to develop fundamental insights into the atomic and molecular dynamics across the electrochemical solid–liquid interface using temporally and spatially resolved synchrotron X-ray spectroscopy, scattering, and imaging. To date, we have made some progress to study the electrochemical interface under operating conditions, through probing metal ion speciation and interfacial structural transformations, as well as determining the atomic and molecular origins of surface-mediated electrochemical processes. Specifically, for batteries we have developed methods to modify the cathode–electrolyte interfacial chemistry through targeted surface doping and symmetry reconstruction. Our studies have established universal design rules for the surface chemistry of intercalating oxides in Li-ion and Na-ion batteries. For water oxidation catalysis, we have discovered that a degraded electrocatalyst can be revivified under catalytic operating conditions by manipulating the reversible phase segregation at the catalyst–electrolyte interface, which constitutes a large space for revivifying and stabilizing catalytic activity . For oxide electrochromics, the interplay between phase transformation, morphological evolution, and nanoscale color heterogeneity results in degraded optical modulation, switching kinetics, Coulombic efficiency, and bleached-state transparency. High interfacial reactivity near the electrode surface is responsible for undesired structural changes of oxide electrochromic materials.
The solid–liquid interfacial reactivity and stability govern the efficiency and lifetime of electrochemical energy devices. Probing and improving the electrochemical solid–liquid interface under operating conditions represents frontier challenges in many aspects of energy sciences, including but not limited to electrocatalytic processes, energy storage and conversion, and chemical synthesis. Our research team has undertaken significant research activities to modulate the electrode surface chemistry for enhancing the interfacial reactivity in batteries and electrocatalysis. Our studies have also found that the high interfacial reactivity triggers undesired reactions with the electrolyte leading to structural changes at the electrode surface and ultimately performance degradation. The mass and charge transfer across the solid–liquid electrochemical interface creates a dynamic environment (e.g., ion concentration) that is sensitive to the electrochemical protocol, electrode surface chemistry, and electrolyte composition. Understanding the interplay between electrode surface chemistry and electrolyte composition will inform the design of better electrochemical interfaces at the atomic and molecular scales. However, probing and tailoring the solid–liquid interfacial processes has ubiquitously remained a key challenge in electrochemical systems. Our long-term goal is to develop fundamental insights into the atomic and molecular dynamics across the electrochemical solid–liquid interface using temporally and spatially resolved synchrotron X-ray spectroscopy, scattering, and imaging. To date, we have made some progress to study the electrochemical interface under operating conditions, through probing metal ion speciation and interfacial structural transformations, as well as determining the atomic and molecular origins of surface-mediated electrochemical processes. Specifically, for batteries we have developed methods to modify the cathode–electrolyte interfacial chemistry through targeted surface doping and symmetry reconstruction. Our studies have established universal design rules for the surface chemistry of intercalating oxides in Li-ion and Na-ion batteries. For water oxidation catalysis, we have discovered that a degraded electrocatalyst can be revivified under catalytic operating conditions by manipulating the reversible phase segregation at the catalyst–electrolyte interface, which constitutes a large space for revivifying and stabilizing catalytic activity . For oxide electrochromics, the interplay between phase transformation, morphological evolution, and nanoscale color heterogeneity results in degraded optical modulation, switching kinetics, Coulombic efficiency, and bleached-state transparency. High interfacial reactivity near the electrode surface is responsible for undesired structural changes of oxide electrochromic materials.
Thrust 3. Oxygen Redox Chemistry in Rechargeable Li-Ion and Na-Ion Batteries
Representative publications: M. M. Rahman, F. Lin* et al. Matter 4, 490 (2021); L. Mu, F. Lin* et al. Nano Lett. 18, 5, 3241 (2018); M. M. Rahman, F. Lin* et al. Nat. Commun. 11, 4548 (2020); M. M. Rahman, F. Lin* et al. Energy Environ Sci. 11, 2496 (2018); M. M. Rahman, F. Lin* et al. ACS Energy Lett. 6, 2882 (2021); X. Zheng, F. Lin* et al. Acta Materialia 212, 116935 (2021); M. M. Rahman, F. Lin* et al. J. Phys. Chem. C 123, 11428 (2019).
Representative publications: M. M. Rahman, F. Lin* et al. Matter 4, 490 (2021); L. Mu, F. Lin* et al. Nano Lett. 18, 5, 3241 (2018); M. M. Rahman, F. Lin* et al. Nat. Commun. 11, 4548 (2020); M. M. Rahman, F. Lin* et al. Energy Environ Sci. 11, 2496 (2018); M. M. Rahman, F. Lin* et al. ACS Energy Lett. 6, 2882 (2021); X. Zheng, F. Lin* et al. Acta Materialia 212, 116935 (2021); M. M. Rahman, F. Lin* et al. J. Phys. Chem. C 123, 11428 (2019).
Background and Scientific Impacts
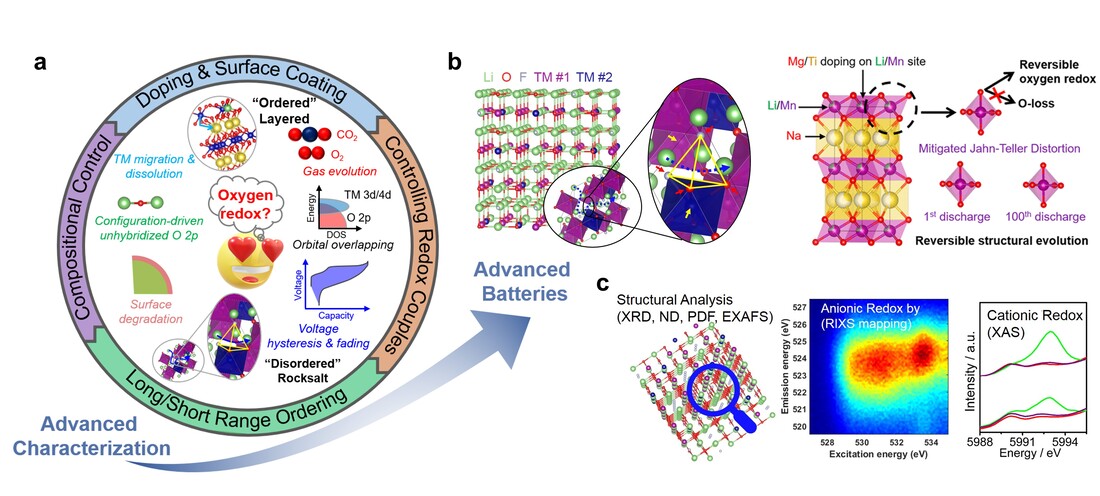
Cationic redox-based oxide cathodes have formed the basis of alkali-ion batteries since the commercialization of Li ion batteries. The limited energy density provided by cationic redox can be overcome by triggering oxygen redox. Hence, oxygen redox has received extensive interest from the perspective of materials development and fundamental understanding. Furthermore, oxygen redox active cathodes can eliminate the use of Ni and Co, the two elements that have raised significant concerns. Although oxygen redox is widely studied, there is still much to understand. Consolidating various concepts of oxygen redox (static/dynamic) can inform a unified understanding. Moreover, materials with oxygen redox create significant challenges such as oxygen evolution, voltage hysteresis/fading, and chemical/structural transformations. A consensus is yet to be achieved on the interrelationship of these phenomena due to the chemical/structural complexities at different length and time scales. We show that local Mn-O symmetry induced structural/chemical evolutions majorly dictate the reversibility of oxygen redox of Na-Li-Mn-O in Na cells. Na-Li-Mn-O with Jahn-Teller distorted MnO6 octahedra undergoes severe Mn dissolution during cycling, which destabilizes the transition metal layer resulting in poor Li retention and irreversible oxygen redox. Jahn-Teller distortion of MnO6 octahedra can be suppressed by modulating the local charge of Mn and Mn-O distance through Mg/Ti dual doping. This leads to reduced Mn dissolution and more reversible oxygen redox. Such stabilization significantly improves the electrochemical performance of Mg/Ti dual doped Na-Li-Mn-O. We show that local structural stabilization through local chemical environment modification can promote reversible oxygen redox in layered cathodes. In a different study, we develop Li-Mn-Cr-O-F (LMCOF) disordered materials through a distinct redox mechanochemical method. The LMCOF contains trivalent Cr and Mn, which allows for simultaneously accessing stable Mn/Cr dual redox at a narrow voltage window acceptable for conventional carbonate electrolytes. Coupled with the mitigated and reversible oxygen chemical changes, the LMCOF delivers high capacity, excellent rate capability, as well as long cycle life upon extensive 1,000 cycles at various current densities.
Cationic redox-based oxide cathodes have formed the basis of alkali-ion batteries since the commercialization of Li ion batteries. The limited energy density provided by cationic redox can be overcome by triggering oxygen redox. Hence, oxygen redox has received extensive interest from the perspective of materials development and fundamental understanding. Furthermore, oxygen redox active cathodes can eliminate the use of Ni and Co, the two elements that have raised significant concerns. Although oxygen redox is widely studied, there is still much to understand. Consolidating various concepts of oxygen redox (static/dynamic) can inform a unified understanding. Moreover, materials with oxygen redox create significant challenges such as oxygen evolution, voltage hysteresis/fading, and chemical/structural transformations. A consensus is yet to be achieved on the interrelationship of these phenomena due to the chemical/structural complexities at different length and time scales. We show that local Mn-O symmetry induced structural/chemical evolutions majorly dictate the reversibility of oxygen redox of Na-Li-Mn-O in Na cells. Na-Li-Mn-O with Jahn-Teller distorted MnO6 octahedra undergoes severe Mn dissolution during cycling, which destabilizes the transition metal layer resulting in poor Li retention and irreversible oxygen redox. Jahn-Teller distortion of MnO6 octahedra can be suppressed by modulating the local charge of Mn and Mn-O distance through Mg/Ti dual doping. This leads to reduced Mn dissolution and more reversible oxygen redox. Such stabilization significantly improves the electrochemical performance of Mg/Ti dual doped Na-Li-Mn-O. We show that local structural stabilization through local chemical environment modification can promote reversible oxygen redox in layered cathodes. In a different study, we develop Li-Mn-Cr-O-F (LMCOF) disordered materials through a distinct redox mechanochemical method. The LMCOF contains trivalent Cr and Mn, which allows for simultaneously accessing stable Mn/Cr dual redox at a narrow voltage window acceptable for conventional carbonate electrolytes. Coupled with the mitigated and reversible oxygen chemical changes, the LMCOF delivers high capacity, excellent rate capability, as well as long cycle life upon extensive 1,000 cycles at various current densities.
Thrust 4. All-Solid-State Li and Na Metal Batteries from Sustainable Resources
Representative publications: D. J. Kautz, F. Lin* et al. J. Mater. Chem. A 6, 16003 (2018); L. Tao, F. Lin* et al. Energy Storage Mater. 24, 129 (2020); Y. Feng, F. Lin* et al. Energy Storage Mater. 31, 274 (2020) ; Y. Feng, F. Lin* et al. J. Mater. Chem. A 7, 26954 (2019) Y. Sun, F. Lin* et al. Nano Lett. 21, 4570 (2021); D. Yu, Lin, F.; Adv. Energy Mater. 2003559 (2021); L. Tao, F. Lin* et al. Adv. Funct. Mater. 30, 2007556 (2021); L. Tao, F. Lin* et al. Adv. Funct. Mater. 30, 2000585 (2021); X. Pan, F. Lin* et al. J Power Sources 403, 127 (2018).
Representative publications: D. J. Kautz, F. Lin* et al. J. Mater. Chem. A 6, 16003 (2018); L. Tao, F. Lin* et al. Energy Storage Mater. 24, 129 (2020); Y. Feng, F. Lin* et al. Energy Storage Mater. 31, 274 (2020) ; Y. Feng, F. Lin* et al. J. Mater. Chem. A 7, 26954 (2019) Y. Sun, F. Lin* et al. Nano Lett. 21, 4570 (2021); D. Yu, Lin, F.; Adv. Energy Mater. 2003559 (2021); L. Tao, F. Lin* et al. Adv. Funct. Mater. 30, 2007556 (2021); L. Tao, F. Lin* et al. Adv. Funct. Mater. 30, 2000585 (2021); X. Pan, F. Lin* et al. J Power Sources 403, 127 (2018).
Background and Scientific Impacts
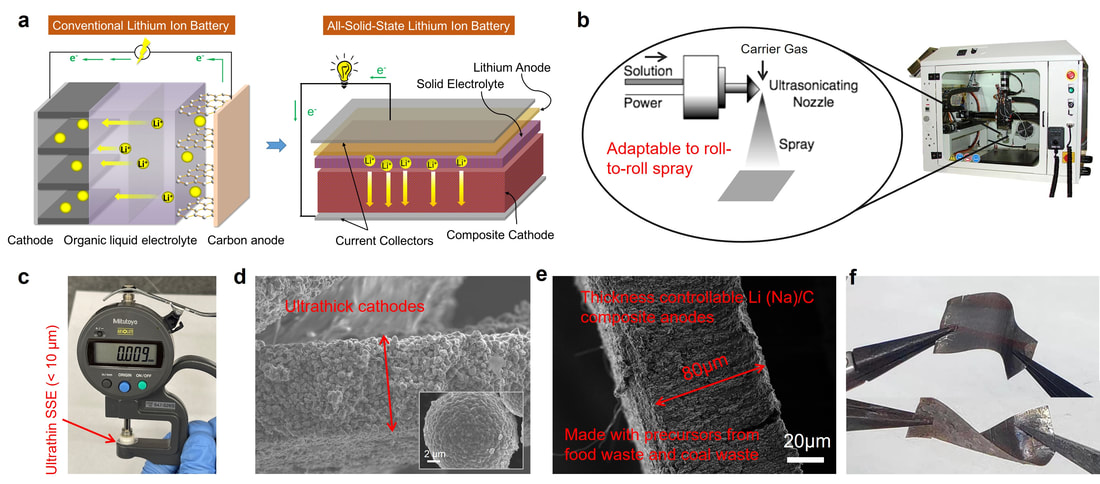
The development of all solid state batteries imposes new challenges for materials electrochemistry and opens up a new dimension to investigate solid-solid interfaces. We have witnessed gradual advancements in developing superionic Li and Na solid electrolytes with favorable bulk conductivity and electrochemical stability windows. The voltage stability quantified by cyclic voltammetry and/or linear scanning voltammetry overestimates the electrochemical stability window of the solid electrolytes, because the functionally important minor interphases (typically a few to tens of nanometers) at Li/solid electrolyte/cathode interfaces are often beyond the detection limit of most traditional electrochemical methods. While these minor interphases have been studied using individual characterization tools, the interfacial electrochemical properties, which are often coupled with the local chemical environments, have not been systematically investigated. Based on our recent success in probing interfaces, such as grain boundaries in cathodes, cathode-liquid electrolyte interfaces, and cathode-polymer electrolyte interfaces, we aim to provide early diagnostics of interfacial changes through advanced chemo-mechanical characterizations, engineered model interfaces, and experimental data-informed predictive neural framework. Our team has accumulated extensive experience in manufacturing large-format solid state cells using the cathode, metal anode, and solid electrolytes. Key scientific challenges reside in the metal anode, including lithium/sodium dendritic growth, low coulombic efficiency, and high interfacial impedance at the lithium/sodium–solid electrolyte interface. Integrating experiments and theoretical modeling, we have designed function-gradient composite metal anodes that offer a protected host for redox reactions and a stable interphase against lithium/sodium consumption.
The development of all solid state batteries imposes new challenges for materials electrochemistry and opens up a new dimension to investigate solid-solid interfaces. We have witnessed gradual advancements in developing superionic Li and Na solid electrolytes with favorable bulk conductivity and electrochemical stability windows. The voltage stability quantified by cyclic voltammetry and/or linear scanning voltammetry overestimates the electrochemical stability window of the solid electrolytes, because the functionally important minor interphases (typically a few to tens of nanometers) at Li/solid electrolyte/cathode interfaces are often beyond the detection limit of most traditional electrochemical methods. While these minor interphases have been studied using individual characterization tools, the interfacial electrochemical properties, which are often coupled with the local chemical environments, have not been systematically investigated. Based on our recent success in probing interfaces, such as grain boundaries in cathodes, cathode-liquid electrolyte interfaces, and cathode-polymer electrolyte interfaces, we aim to provide early diagnostics of interfacial changes through advanced chemo-mechanical characterizations, engineered model interfaces, and experimental data-informed predictive neural framework. Our team has accumulated extensive experience in manufacturing large-format solid state cells using the cathode, metal anode, and solid electrolytes. Key scientific challenges reside in the metal anode, including lithium/sodium dendritic growth, low coulombic efficiency, and high interfacial impedance at the lithium/sodium–solid electrolyte interface. Integrating experiments and theoretical modeling, we have designed function-gradient composite metal anodes that offer a protected host for redox reactions and a stable interphase against lithium/sodium consumption.